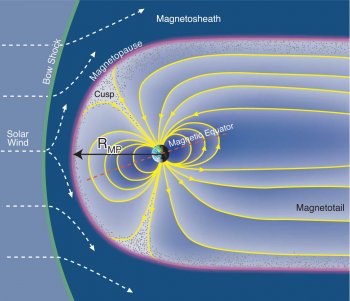
I have no idea what you are trying to say here.
The ions in the ionosphere are produced by photolysis of the upper atmosphere by UV and other high energy solar radiation
"Ionization at any given level depends on three factors—the availability of photons of a wavelength capable of effecting ionization, a supply of atoms and molecules necessary to intercept this
radiation, and the
efficiency with which the atoms and molecules are able to do so
The efficiency is relatively large for O, O2, and N2 from about 10 to 80 nm. This is the portion of the spectrum responsible for production of electrons and ions in the F1 region.
Photons with wavelengths between 90 and 100 nm are absorbed only by O2. They therefore penetrate deeper and are responsible for producing about half the ionization in the E layer. The balance is derived from so-called “soft”
X-rays (those of longer wavelengths), which are absorbed with relatively low efficiency in the
F region and so are able to penetrate to
altitudes of about 120 km (75 miles) when the
Sun is high over the region. “Hard” X-rays (those of shorter wavelengths—that is, below about 5 nm) reach even deeper. This portion of the spectrum accounts for the bulk of the ionization in the D region, with an additional contribution from wavelengths longer than 102.6 nm—mainly from photons in the strong solar emission line at Lyman α at a wavelength of 121.7 nm. (The
Lyman series is a related sequence of wavelengths that describe electromagnetic energy given off by energized atoms in the ultraviolet region.) Lyman α emissions are weakly absorbed by the major components of the atmosphere—O, O2, and N2—but they are absorbed readily by NO and have sufficient energy to ionize this relatively unstable
compound. Despite the low abundance of NO, the high flux of
solar radiation at Lyman α is able to provide a significant source of ionization for the D region near 90 km (55 miles)."
ionosphere and magnetosphere - Mechanisms of ionization
Pax et Bonum